
Bookmark This Page
| Daily Dividend Report: IBM,WMB,HON,LIN,GPC
Tue, 30 Apr 16:26:22 GMT |
| Tuesday Sector Laggards: Education & Training Services, Precious Metals
Tue, 30 Apr 16:21:29 GMT |
   
|
|
Get a quote box (like the one below) for your site!
|
energy quotes gold quotes
uranium stocks
solar power stocks
wind power stocks
Industry focus:
advertising stocks,
space stocks,
aerospace stock,
aerospace sector,
list of aerospace companies,
largest chemical companies,
chemical stock,
chemical news,
best agriculture stocks,
ag stocks,
chinese agriculture stocks,
top agriculture companies,
agriculture stocks,
agricultural stocks,
agricultural stock,
stocks agriculture,
agriculture markets,
agriculture index,
agriculture industries,
agricultural investment,
agriculture investment,
agricultural industry,
farm stock,
airline stock symbols,
airline stock prices,
airline stock,
airlines stock,
clothing stock,
fashion stocks,
publicly traded fashion companies,
clothing company stocks,
apparel stock,
apparel companies,
application software stocks,
asset management stocks,
auto stocks,
auto industry stocks,
chinese auto stocks,
auto stock prices,
automotive stock,
auto parts stocks,
community bank stocks,
regional bank stocks,
canadian bank stocks,
banking stock,
national bank stocks,
commercial bank stock,
banks stock,
bank stock quote,
bank stocks,
banking industry,
alcohol stocks,
beverage stock,
global wine stocks,
wine stocks,
liquor stock,
biotech stocks list,
biotechnology investing,
public biotech companies,
top biotech stocks,
nanotechnology stock,
largest biotech companies,
biotechnology stock,
biotech investing,
investing in biotech,
best biotech companies,
bio stocks,
biotech sector,
biotechnology investment,
biopharma companies,
new biotech companies,
biotech investment,
biotechnology industries,
nanotech stocks,
biotech stocks,
biotechnology articles,
biotechnology news,
business stocks,
service stocks,
chemical companies,
chemical industries,
chemical industry,
chemical company,
chemicals company,
cigarette stock,
cigarette company stocks,
cigarette stock symbols,
tobacco company stocks,
tobacco stock,
cigar stocks,
communications stocks,
communication stock,
computer peripherals companies,
computer peripherals,
computers stocks,
computer stock,
computer web,
internet stocks,
construction stocks,
machinery stocks,
builders stocks,
building stocks,
consumer goods stocks,
consumer services stocks,
consumer services companies,
lending stocks,
mortgage banking,
lending companies,
mortgage bankers,
loan services,
mortgage services,
mortgage bank,
loan bank,
defense stocks,
defensive stock,
department store stocks,
diagnostic company,
diagnostic companies,
pharmaceuticals stocks,
drug stocks,
drug company stocks,
pharma stock,
education stocks,
college stock,
electric utility stocks,
electric company stocks,
electric utilities stocks,
utility stocks,
utilities stocks,
power equipment companies,
electrical supply companies,
electronic stocks,
entertainment stock,
movie stocks,
movies companies,
movie company,
cefs,
open ended and closed ended mutual funds,
closed ended investment,
closed ended fund,
bonds fund,
closed end,
food stock,
game stock,
gambling stocks,
casino stocks,
gaming stocks list,
gaming stocks,
gas utility companies,
gas company stocks,
construction industries,
builders contractors,
construction services,
construction industry,
grocery store stocks,
supermarket stock,
drug store stocks,
home stocks,
furniture stock,
home improvement stocks,
medical company stocks,
top medical stocks,
medical stock,
hospital stock,
medical supply stocks,
medical technology stocks,
medical device stocks,
medical equipment stocks,
copper mining,
palladium mining stocks,
mining metals,
mining,
mining news,
gold exploration,
mining share price,
lithium mines,
mining industries,
international mining companies,
mining information,
molybdenum mining companies,
nickel mining companies,
metals and mining stocks,
gold and silver mining stocks,
copper mining companies,
rare earth mining companies,
rare metals stocks,
rare earth stocks,
metals stocks,
welding stock,
nonprecious metals,
non metallic mining,
office supplies companies,
office supply companies,
oil services stocks,
oil pipeline stocks,
gas pipeline stocks,
gas pipeline companies,
pipeline companies,
natural gas pipeline companies,
oil services companies,
oil field services,
oil service stocks,
natural gas pipelines,
oilfield service companies,
oil and gas pipeline companies,
oil gas pipeline,
oil exploration stocks,
oil exploration sector,
oil exploration companies,
oil drilling stocks,
oil drilling companies,
oil production companies,
china oil companies,
brazil oil companies,
china oil stocks,
brazil oil stocks,
oil companies,
oil stocks,
oil drilling,
oil exploration,
offshore oil drilling companies,
list of oil drilling companies,
oil and gas exploration,
oil and gas drilling,
oil and gas stocks,
oil and gas drilling companies,
oil refining companies,
oil marketing companies,
oil refining stocks,
oil refining sector,
oil refinery companies,
oil refinery stocks,
major oil companies,
oil sector,
oil refinery,
oil refinery company,
oil company,
oil marketing company,
oil refining company,
oil refining industry,
major oil companies list,
oil and gas companies,
crude oil stocks,
packaging companies,
container companies,
packaging stocks,
packaging sector,
container sector,
pulp stocks,
paper stocks,
timber stocks,
pulp companies,
paper companies,
timber companies,
timber trusts,
cardboard companies,
paper sector,
timber sector,
paper companies list,
silver mining companies,
gold mining companies,
gold mining sector,
precious metal stocks,
mining companies,
exploration sector,
mining sector,
exploration stocks,
mining stocks,
silver stocks,
gold stocks,
gold mining stocks,
silver mining stocks,
silver mining company,
canadian mining companies,
gold mining,
gold mining company,
mining company,
list of mining companies,
gold stocks list,
largest gold mining companies,
silver mining,
printing companies,
printing stocks,
printing sector,
newspaper stocks,
newspaper sector,
newspaper companies,
publishing stocks,
publishing sector,
publishing companies,
digital media companies,
digital media stocks,
digital media sector,
book publishing companies,
digital media company,
publishing company,
railroad stocks,
railroad sector,
railroad companies,
railroad company,
railroad investment,
major railroad companies,
real estate companies,
real estate stock,
real estate public companies,
real estate investing,
real estate investments,
real estate sector,
commercial real estate investing,
real estate investment firms,
real estate investing guide,
REITs,
real estate investment trust,
REIT sector,
REIT stocks,
REITs sector,
REITs stock,
public REITs,
real estate investment trusts,
real estate investment trust companies,
real estate investment trusts REITs,
real estate investment companies,
real estate investment company,
real estate investment trust REIT,
rubber stocks,
plastic stocks,
rubber companies,
plastic companies,
rubber sector,
plastic sector,
plastic manufacturing companies,
rubber company,
plastic company,
semiconductor stocks,
semiconductor investments,
semi stocks,
semiconductor companies,
semiconductor sector,
shipping stocks,
dry bulk stocks,
container stocks,
dry bulk shipping,
dry bulk shipping companies,
tanker stocks,
shipping companies,
shipping sector,
specialty retail,
retail stocks,
retail investing,
retail store stocks,
consumer stocks,
consumer investment,
retail companies,
retail sector,
sports stocks,
sports investing,
sporting goods stocks,
sports investments,
sporting goods companies,
sporting goods sector,
stock message boards,
television stocks,
television investment,
radio stocks,
radio invest,
media stocks,
media invest,
media investment,
media investing,
television companies,
television sector,
radio sector,
radio companies,
media companies,
media sector,
textile stocks,
apparel stocks,
textile investment,
textile companies,
textile sector,
apparel sector,
freight investment,
transportation investment,
truck investment ,
freight stocks,
transportation stocks,
trucking stocks,
trucking companies,
trucking sector,
waste management stocks,
waste stocks,
recycling stocks,
waste investment,
waste companies,
waste sector,
water stocks,
water utilities,
water investing,
water investment,
water companies,
water sector
Theralase(R) Provides Update on Phase II Bladder Cancer Clinical StudyTo date, Study II has provided the primary study treatment for 63 patients. In 2016, Kamat et al. stated in the Journal of Clinical Oncology that the International Bladder Cancer Group (" IBCG ") recommended that, " Single-arm designs may be relevant for the BCG-unresponsive population. Here, a clinically meaningful initial complete response rate (for carcinoma in situ) or recurrence-free rate (for papillary tumors) of at least 50% at 6 months, 30% at 12 months and 25% at 18 months is recommended. " [1] Based on the 63 patients treated to date, the interim clinical data for Study II is presented below:
The Study II interim clinical data demonstrates a Complete Response (" CR ") of 54% at 6 months, 38% at 12 months and 37% at 15 months, which exceeds the IBCG guidelines. In addition, the Study II interim clinical data demonstrates that at the 90 Day Assessment Visit, 56% of Evaluable Patients achieved a CR and 63% achieved a Total Response (CR + IR), while at 450 days, 37% achieved a CR and 41% achieved a TR. 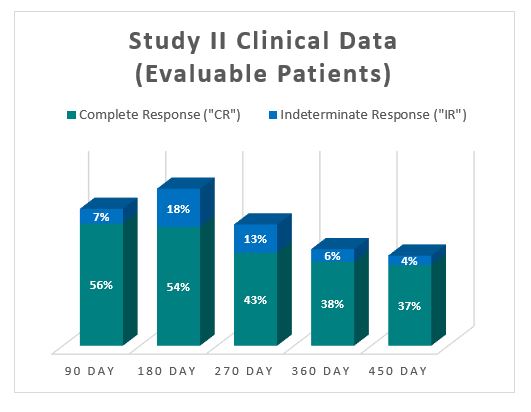 For evaluable patients in Study II, who received the optimized Study II Treatment (Post August 1, 2020), the interim clinical data is presented below:
The interim clinical data for patients who received the optimized Study II Treatment demonstrates that at the 90 Day Assessment Visit, 62% of Evaluable Patients achieved a CR and 68% achieved a Total Response (CR + IR), while at 450 days, 39% achieved a CR and 44% achieved a TR. 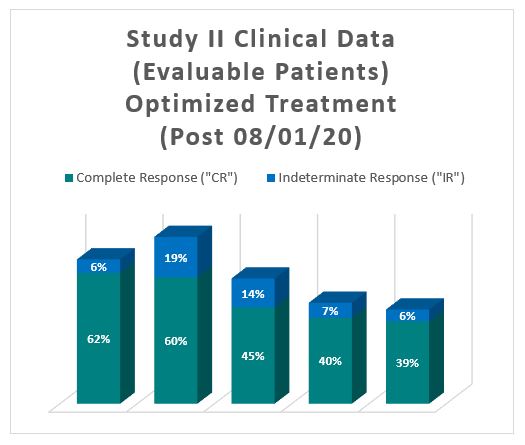 Notes:
The Swimmer's plot below is a graphical representation of the interim clinical results (n=63) graphically demonstrating a patient's response to a treatment over time. As can be seen in the plot, clinical data is still pending for patients, who have demonstrated an initial CR at 90 days and continue to demonstrate a duration of that response. 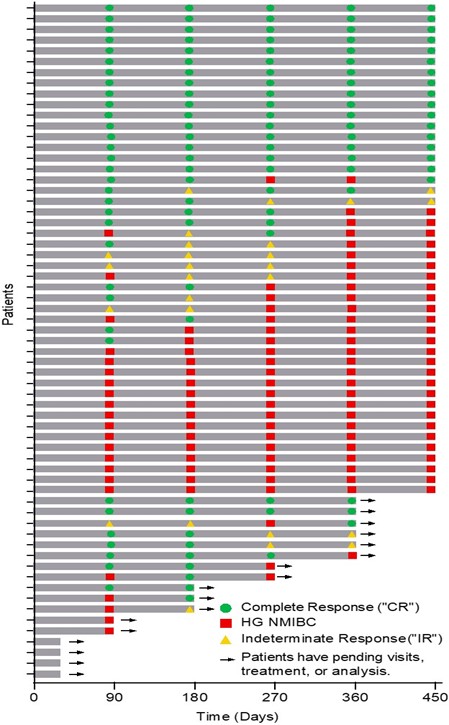 The Swimmer's Plot illustrates:
The Kaplan-Meier (" KM ") Curve below represents the interim cumulative incidence of clinical events; including, the treatment efficacy occurring over a prespecified time in Study II. 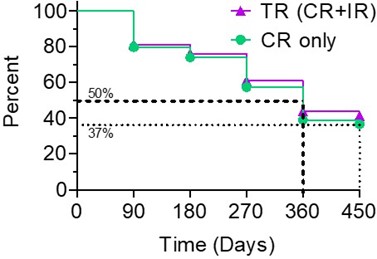 According to the interim clinical data in the KM curve:
For 63 patients treated in Study II, there have been 13 Serious Adverse Events (" SAEs ") reported:
Theralase believes all SAEs reported to date are unrelated to the Study II Drug or Study II Device. In 2020, the FDA granted Theralase Fast Track Designation (" FTD ") for Study II. As a Fast Track designee, Theralase has access to early and frequent communications with the FDA to discuss Theralase 's development plans and ensure the timely collection of clinical data to support the approval process. The accelerated communication with the FDA potentially allows, the Study II Treatment, to be the first intravesical, patient-specific, light-activated, Ruthenium-based PDC for the treatment of patients diagnosed with BCG-Unresponsive NMIBC CIS, (with or without recurrent / resected papillary T a /T 1 tumours). FTD can lead to Break Through Designation (" BTD "), Accelerated Approval (" AA ") and/or Priority Review, if certain criteria are met. In mid-2023, the Company submitted a pre-BTD submission to the FDA and based on the FDA's feedback, the Company is currently working with the Clinical Study Sites (" CSSs "), a central pathology organization, a biostatistics organization and a regulatory consulting organization to update the pre-BTD with clinical data clarifications identified by the FDA. The Company plans to resubmit the pre-BTD submission to the FDA in 1Q2024 for FDA review of these clarifications. Once the pre-BTD submission has been accepted by the FDA, the Company plans to compile a BTD submission for review by the FDA in support of the grant of a BTD approval. Theralase is working to complete enrollment and delivery of the primary Study II Treatment for all patients in 2024. If successful, this would allow clinical data lock in mid-2026 with a potential Health Canada and FDA approval by 2026 / 2027. About Study II: Study II utilizes the therapeutic dose of the patented Study II Drug (" Ruvidar TM " or " TLD-1433 ") (0.70 mg/cm 2 ) activated by the proprietary Study II Device (" TLC-3200 Medical Laser System "). Study II is focused on enrolling and treating approximately 100 BCG-Unresponsive NMIBC Carcinoma In-Situ (" CIS ") patients in up to 15 CSSs located in Canada and the United States. About Ruvidar TM : Ruvidar TM is a patented PDC with 12 years of published peer reviewed preclinical research and is currently under investigation in Study II. About Theralase Technologies Inc.: Theralase is a clinical stage pharmaceutical company dedicated to the research and development of light activated compounds, their associated drug formulations and the light systems that activate them, with a primary objective of efficacy and a secondary objective of safety in the destruction of various cancers, bacteria and viruses. Additional information is available at www.theralase.com and www.sedar.com Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release. Forward Looking Statements: This news release contains "forward-looking statements" within the meaning of applicable Canadian securities laws. Such statements include; but are not limited to statements regarding the Company's proposed development plans with respect to Photo Dynamic Compounds and their drug formulations. Forward looking statements may be identified by the use of the words " may , " should ", " will ", " anticipates ", " believes ", " plans ", " expects ", " estimate ", " potential for " and similar expressions; including, statements related to the current expectations of Company's management for future research, development and commercialization of the Company's Photo Dynamic Compounds and their drug formulations, preclinical research, clinical studies and regulatory approvals. These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to: adequately fund, and secure the requisite regulatory approvals to successfully complete a Phase II NMIBC clinical study in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its drug formulations, the risk that access to sufficient capital to fund the Company's operations may not be available or may not be available on terms that are commercially favorable to the Company, the risk that the Company's drug formulations may not be effective against the diseases tested in its clinical studies, the risk that the Company's fails to comply with the term of license agreements with third parties and as a result loses the right to use key intellectual property in its business, the Company's ability to protect its intellectual property, the timing and success of submission, acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict. Readers should not unduly rely on these forward- looking statements which are not a guarantee of future performance. There can be no assurance that forward looking statements will prove to be accurate as such forward looking statements involve known and unknown risks, uncertainties and other factors which may cause actual results or future events to differ materially from the forward-looking statements. Although the forward-looking statements contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these forward-looking statements. All forward-looking statements are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such statements. For More Information: 1.866.THE.LASE (843-5273) Kristina Hachey, CPA [1]Kamat AM et al. J Clin Oncol. 2016; 34: 1935-1944 SOURCE: Theralase Technologies Inc. View the original press release on accesswire.com
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||